Standing in the fertilizer aisle of a garden or farm store, you are faced with a dizzying array of fertilizer options, many with a series of three numbers like 10-10-10, 20-20-20, 10-8-10 or many other combinations of numbers.
You may be asking yourself, “What do the numbers on fertilizer mean?”. These are NPK values, which leads to the next question of, “What is NPK?”. Keep reading to learn more about fertilizer numbers and NPK.
What Do the Numbers on Fertilizer Mean?
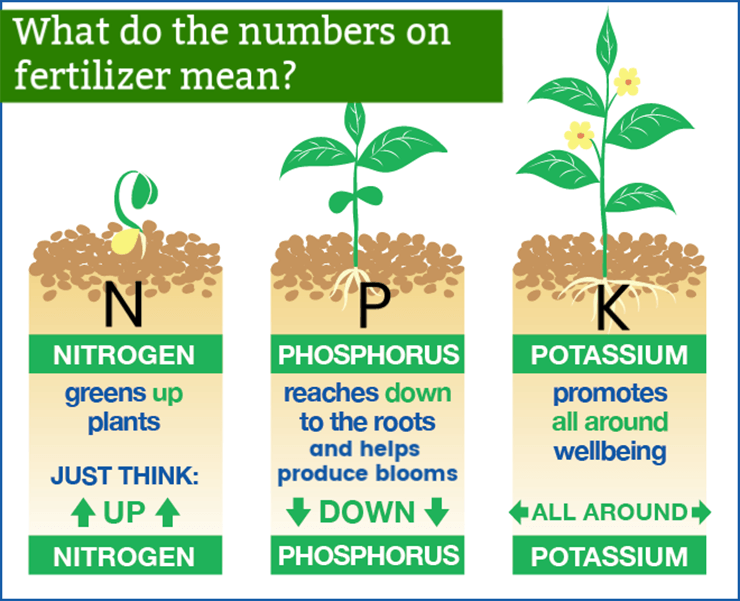
The three numbers on fertilizer represents the value of the three macro-nutrients used by plants. These macro-nutrients are nitrogen (N), phosphorus (P), and potassium (K), or NPK for short.
The higher the number, the more concentrated the nutrient is in the fertilizer. For example, numbers on fertilizer listed as 20-5-5 has four times more nitrogen in it than phosphorus and potassium.
A 20-20-20 fertilizer has twice as much concentration of all three nutrients than 10-10-10. The fertilizer numbers can be used to calculate how much of a fertilizer needs to be applied to equal 1 pound (453.5 g.) of the nutrient you are trying to add to the soil. So if the numbers on the fertilizer are 10-10-10, you can divide 100 by 10 and this will tell you that you need 10 pounds (4.5 kg.) of the fertilizer to add 1 pound (453.5 g.) of the nutrient to the soil. If the fertilizer numbers were 20-20-20, you divide 100 by 20 and you know that it will take 5 pounds (2 kg.) of the fertilizer to add 1 pound (453.5 g.) of the nutrient to the soil. A fertilizer that contains only one macro-nutrient will have “0” in the other values. For example, if a fertilizer is 10-0-0, then it only contains nitrogen.
What is NPK and Why is it Important?
So now that you know what the numbers on fertilizer mean, you need to know why NPK is important to your plants. All plants need nitrogen, phosphorus, and potassium to grow. Without enough of any one of these nutrients, a plant will fail.
Nitrogen (N) – Nitrogen is largely responsible for the growth of leaves on the plant.
Phosphorus (P) – Phosphorus is largely responsible for root growth and flower and fruit development.
Potassium (K) – Potassium is a nutrient that helps the overall functions of the plant perform correctly. Knowing the NPK values of a fertilizer can help you select one that is appropriate for the type of plant you are growing.
For example, if you are growing leafy vegetables, you may want to apply a fertilizer that has a higher nitrogen number to encourage leafy growth. If you are growing flowers, you may want to apply a fertilizer that has a higher phosphorus number to encourage more blooms.
Before you apply fertilizer to your garden beds, you should have your soil tested. This will also help you determine what balance of fertilizer numbers will be appropriate for your garden’s soil needs and deficiencies.




